WHAT ARE VACCINES AND HOW DO THEY WORK
A vaccine consists of pathogens, which are micro-germs (microbes- bacteria or virus) in killed (inactive) or weakened (attenuated) form, or as their parts or genetic material, which on entering the human body, help produce immunity against that particular infection without actually suffering from it.
Our body’s immune system can recognize specific parts of these pathogens called ‘antigens’, and in response produce ‘antibodies’ and activate immune cells with memory, which can destroy these pathogens. The body’s immune system takes some time in the process of recognition, activation, and initiation of such a specific adaptive immune response. In this window period, the invading microbe can rapidly multiply and overwhelm the immune system, thereby producing symptoms and causing damage. However, if vaccinated, the body’s immune system is ready to immediately fight off the actual active organism when exposed, significantly lowering the risk of getting the disease and preventing severe disease or its complications.
Vaccination is the process of giving a vaccine while immunization refers to the body developing immunity against that particular infection. If the person is vaccinated and does not develop an immune response with the formation of antibodies, then the person is not immunized. Inoculation refers to introducing microbes into any medium, and in humans, is synonymous with vaccination.
TYPES OF VACCINES
Vaccines have conventionally used either the whole or a part (containing the antigens) of the disease-causing pathogen for immunization. Now new types of vaccines using genetic technology have been developed during the COVID pandemic and are now available in many countries.
Whole Microbe Vaccines
Killed (Inactivated)
These vaccines contain the whole organism but are killed and inactivated by heat or chemicals so that they cannot cause any disease, but the antigens can be recognized by the body’s immune system. Examples are injectable vaccines for influenza (flu), whole cell pertussis (whooping cough), hepatitis A, polio, and rabies. The COVID vaccines Covaxin (Bharat Biotech-ICMR-NIV India) and the vaccines from Sinovac/Sinopharm China, are also killed vaccines.
Live weakened
Also called live attenuated, these vaccines contain the virus in a highly weakened form, therefore, incapable of causing disease (or sometimes causing extremely mild symptoms). Examples are the injectable vaccines for tuberculosis (BCG), measles, mumps, rubella (MMR), chickenpox (Varicella), and oral liquid vaccine for rotavirus, along with the oral (mouth drops) vaccine for polio, oral capsule vaccine for typhoid, and the nasal spray vaccine for flu.
Killed-inactivated versus Live-weakened Vaccines:
The advantage of giving a live vaccine is that since the organism can multiply in the body, the immune response and antibody production are much greater and long-lasting, as compared to killed vaccines, and therefore, additional booster doses may not usually be needed. Sometimes in killed vaccines, substances called adjuvants (like aluminum salts) are added to further enhance the immune response.
Live vaccines are more difficult to develop and manufacture, and they also require more stringent storage and cold chain. Live vaccines are to be avoided in people with compromised or suppressed immunity, in whom killed vaccines can be safely given. Extremely rare cases of live weakened pathogens mutating to a stronger infective (virulent) form have been reported.
Part Microbe Vaccines
Subunit vaccines
Instead of the whole organism, a part of it, like a protein or polysaccharide subunit (containing the antigens), is given to produce the desired immune response. These subunits can be isolated from the organism by chemical disintegration or prepared in the laboratory by genetic recombinant technology. Like killed vaccines, subunit vaccines are also safe in people with weakened immune systems. They may contain adjuvants to enhance immune response, but since only the antigens are being given, instead of the whole organism, the immune response may be more efficient (precise immune targeting).
Examples of protein subunit vaccines are acellular pertussis (aP), recombinant hepatitis B and herpes zoster (shingles), human papillomavirus (HPV), and the COVID protein subunit vaccines Corbevax (India) and Novovax-Nuvaxovid (Covovax-India). Examples of polysaccharide subunit vaccines include the pneumococcal (PPSV23) and meningococcal (Men-ACWY) vaccines.
Conjugate vaccines
It is a type of subunit vaccine where a weak antigen (a polysaccharide) is combined with a strong antigen (a protein or toxoid) as a carrier and marker so that the immune system generates a stronger response to the weak antigen.
An example is the pneumococcal conjugate vaccine (PCV) that is given to infants and children (as they have an immature immune system) and to adults with associated medical conditions and lowered immunity. Similarly, the meningococcal ACWY vaccine consists of polysaccharide subunits conjugated with the diphtheria toxoid. Others include Hemophilus influenzae (Hib) and typhoid conjugate vaccines.
Some organisms have multiple variants (serotypes), so the vaccine would need to have antigens from all important serotypes to confer effective protection. For example, the pneumococcal conjugate vaccines PCV10, PCV13, and now available PCV20, the pneumococcal polysaccharide vaccine (PPSV23), the trivalent and tetravalent flu vaccines, and bivalent, tetravalent, and now nonavalent HPV vaccines.
Toxoid vaccines
These consist of inactivated toxins (toxoids) released by certain bacteria that cause the disease, and are recognized by our immune system. Examples are vaccines for diphtheria and tetanus.
Combining vaccines
Combination vaccines of toxoids and killed vaccines (DPT, DTwP, Tdap) or multiple live vaccines (MMR and MMRV- measles, mumps, rubella, varicella) are available for more efficient immunization, especially in children.
The pentavalent vaccine gives protection from diphtheria, tetanus, pertussis, hepatitis B, and Haemophilus influenzae type b in a single vaccine, while the hexavalent vaccine also adds protection from polio.
Routes of Vaccination
Most vaccines are given as injections; however, some may be given orally (live typhoid and polio vaccines) or nasally (live flu vaccine). These routes give the advantage of stimulating effective local immunity in the gut or respiratory tract, which are the entry routes of the pathogens.
In some cases, for the same infection, different types and routes of vaccines may be available, as seen with typhoid (subunit/conjugate injectable and live oral), polio (killed injectable and live oral), and flu (killed injectable and live nasal).
Vaccine recommendations in different populations
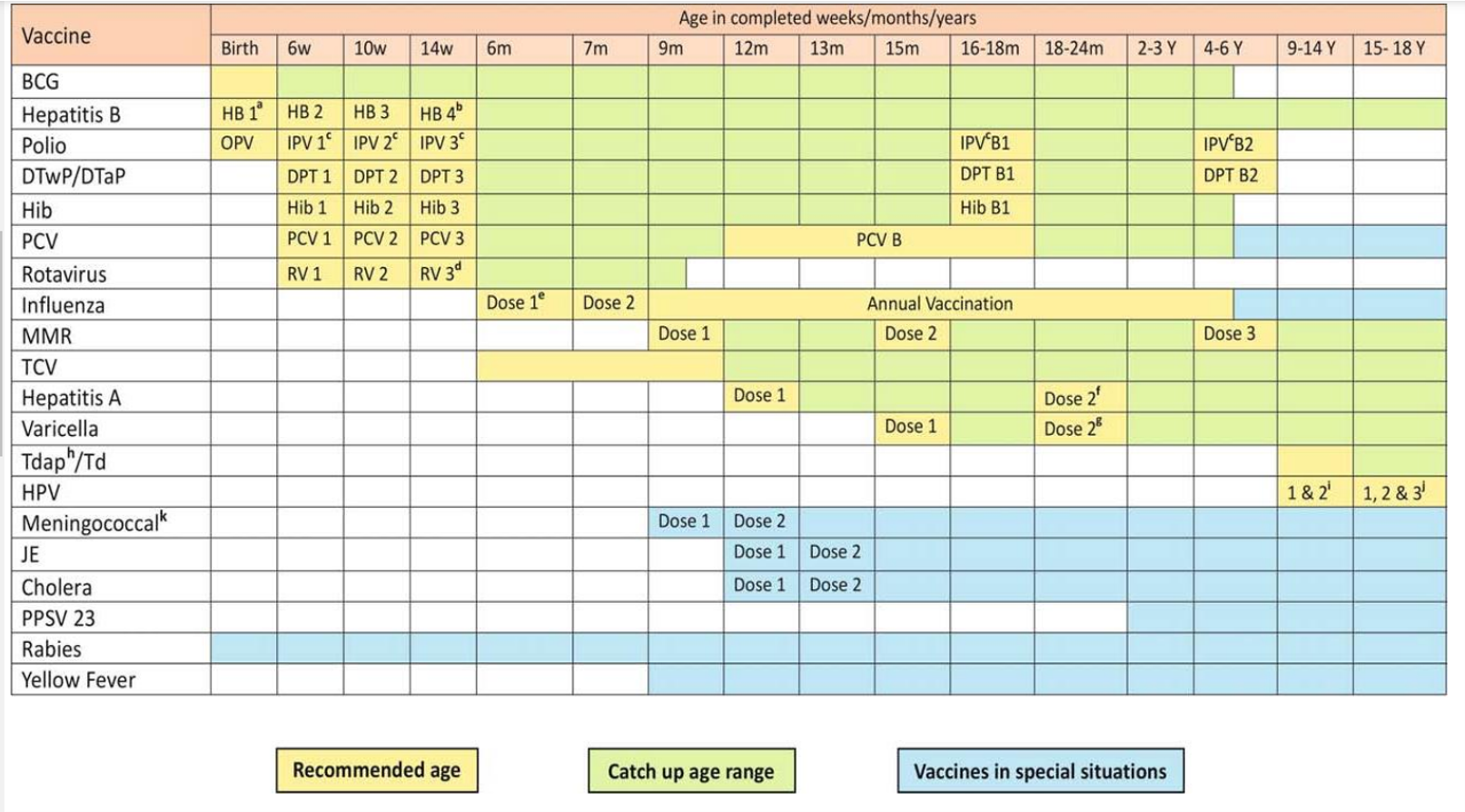
PEDIATRIC VACCINATION RECOMMENDATIONS


ADULT VACCINATION RECOMMENDATIONS
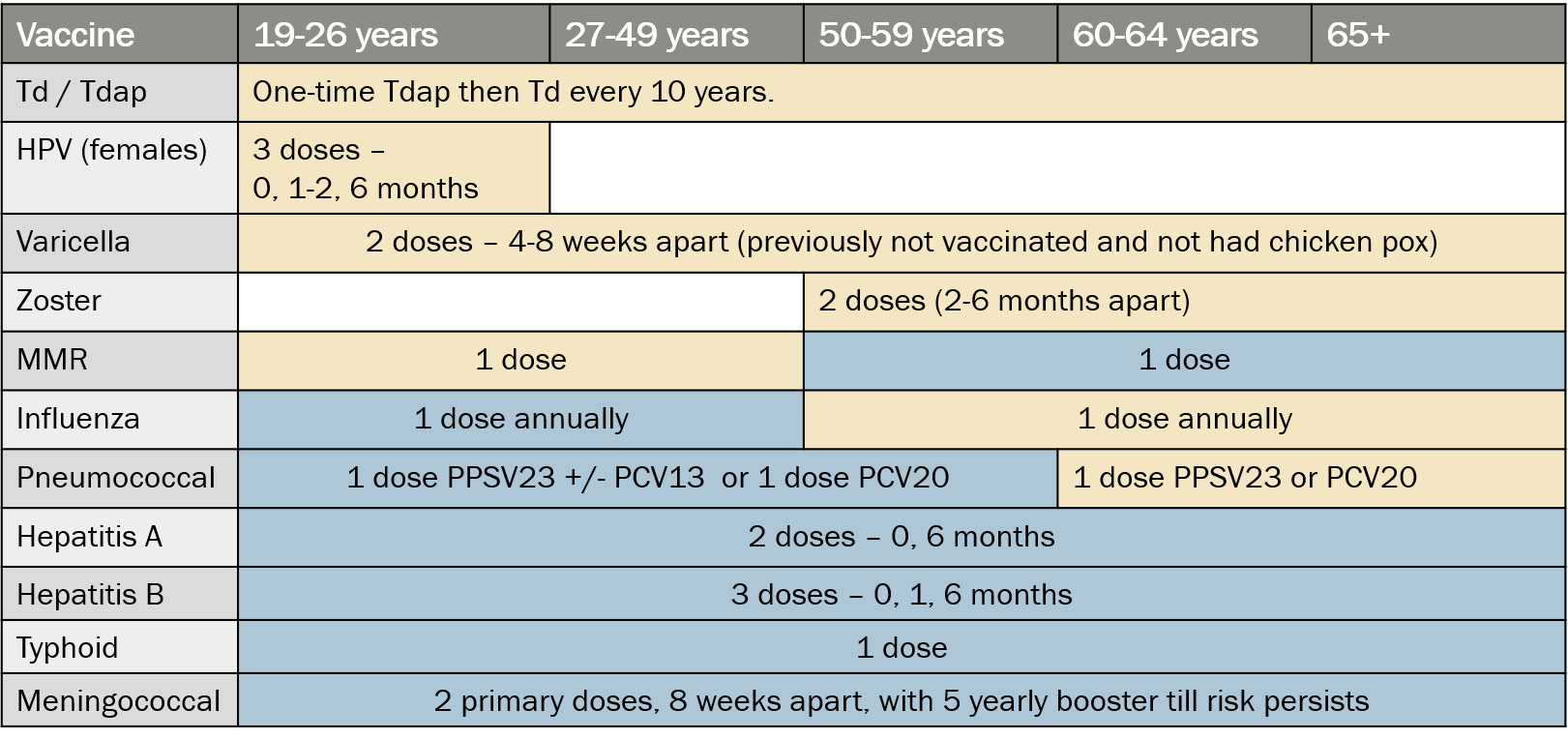


In the above tables, vaccines marked in yellow are given to all adults who fit the age and medical criteria. The vaccines marked in blue are only for high-risk groups like those with medical co-morbidities including cardiovascular disease, diabetes, chronic lung disease, kidney and liver dysfunction, conditions causing compromised immunity (HIV, other immunodeficiencies, absence of spleen, organ transplant, immunosuppressive drugs), or presence of lifestyle or travel related, or occupational risk to the particular infection.
In India, Td/Tdap and Flu vaccine doses are mandated in pregnancy irrespective of previous vaccination status. All other vaccines should be given either a month or more before planning pregnancy or after delivery. All live vaccines are contraindicated in pregnancy.
COVID vaccine recommendations are updated periodically according to country-wise and global status of cases, spread, and variants.
New Types of Vaccines
Using the genetic material of viruses to produce the required antigen inside the human body has heralded a new age in vaccine technology. This is like setting up factories inside our bodies to produce the desired antigen in large amounts to generate effective and more lasting immunity. The immune response consists not only of antibody production (humoral immunity) but also of immune cells causing direct destruction of the viral antigen (T cell or cell-mediated immunity).
Vaccines for COVID have made use of these new technologies which enable a much faster and more large-scale production, which made availability for clinical trials and public immunization possible during the pandemic. These vaccines do not contain any live virus so cannot cause the disease and are also safe in people even with possible immune suppressive conditions.
Adenoviral vector vaccines
The genes of the virus for the required antigen (surface spike protein in case of coronavirus), are loaded into another virus (vector) like the adenovirus which is rendered replication-incompetent (non-replicating), and then introduced into the human body. The body will produce antibodies and an immune response against the specific antigen as well as the vector. Therefore if the vector is a virus that humans are routinely exposed to, then there might be pre-existing antibodies against it which can destroy the vector before the viral genes can produce good amounts of required antigens. This can be prevented by using vectors that have low exposure in humans or mainly infect animals.
Examples of such vaccines approved are the Oxford-Astra Zeneca COVID vaccine using an adenoviral cold virus of chimpanzees, the Sputnik V vaccine by Gamaleya, Russia which uses two human adenoviral strains (Ad5 and 26), and the vaccine from Johnson and Johnson using the Ad26 vector. A chimpanzee adenoviral vector has also been used in the nasal COVID vaccine developed in India. Before the COVID pandemic, the adenoviral vector vaccine technology has already been evaluated in humans against the Ebola virus and malaria.
mRNA vaccines
Genetic information is transferred (transcripted) from the main viral DNA or RNA to a messenger RNA (mRNA) which actually produces the antigen proteins by a process called translation. Injecting the mRNA containing the transcripted genetic information can produce the desired viral antigenic protein in our body. This technology enables the manufacturing of large quantities of vaccines quickly.
Examples are the COVID vaccines by Moderna, Pfizer-BioNtech, and Gennova mRNA vaccines.
While mRNA vaccines are being used for the first time in humans during COVID, they have been used successfully in veterinary practice and also evaluated in humans in the past for Zika virus.
Plasmid DNA vaccine
The genes containing information to make the required viral antigen are inserted into the DNA of molecules called plasmids, found in bacteria. These plasmids are then introduced into the human body.
The COVID vaccine from Zydus-Cadila called ZyCoV-D has been developed with this technology in India.
HERD IMMUNITY
Herd immunity is a form of indirect protection from a particular infection when a sufficient percentage of a population has become immune to an infection, either through vaccination or exposure to the infection itself. This reduces the likelihood of infection for individuals who have not been immunized or exposed to the infection, due to the overall lowered spread of the infection in the community. This can happen after more than 50-60% population is exposed to the infection or immunized against it.
SIDE EFFECTS AND COMPLICATIONS OF VACCINATION
Any medicine, treatment, or vaccine can have side effects. All vaccines are thoroughly tested for safety in clinical trials before they enter the market for general population use.
Common
In the majority of cases, vaccination produces none or minimal side effects like transient pain or swelling at the injection site, fatigue, weakness, headache, muscle/joint pains, chills, or fever. These symptoms seldom last more than a day or two, and usually do not need any treatment, though sometimes one or two doses of medicines for pain or fever may be prescribed.
Some people may feel faint, dizzy, or experience blurring and ringing in the ears, after any medical procedures, including vaccination. Sitting or lying down for about 15 minutes can help prevent fainting and injuries caused by a fall.
Rare
Allergic reactions that can sometimes be serious (called anaphylaxis) can occur rarely after vaccination. Though this is seen mainly in people who have a history of allergic reactions, it can also occur unexpectedly. It has in some cases been linked to the presence of substances like polyethylene glycol (PEG) or polysorbates in vaccines. The symptoms can range from mild localized itching, redness, and swelling, to itching all over the body with hives, swelling of the lips, sweating, palpitation, and breathlessness. These reactions can be managed with adrenaline (epinephrine) and hydrocortisone injection in severe cases, and with antihistamine medicines in milder allergic reactions. Often people with a history of allergies carry such medicines with them, and these are available at vaccination centers.
Neurological side effects have also been very rarely reported with vaccines, which can be seen even a few days or weeks after taking the vaccine. These include Bell’s palsy, Guillain-Barre syndrome, transverse myelitis, multiple sclerosis, meningitis, encephalitis, and seizures. Though worrisome as it may sound, these are extremely uncommon, and most of these cases recover well.
Thrombosis, which is the formation of clots in blood vessels is an extremely rare but serious and sometimes fatal side effect. Sometimes these clots can detach from the vessel wall and travel (emboli). Clots and emboli can hamper blood supply to vital organs like the lungs (pulmonary embolism), brain (stroke), heart (heart attack), etc, causing complications and rarely death. The development of thrombosis post-vaccination is sometimes referred to as vaccine-induced immune thrombotic thrombocytopenia (VITT).
References
Vaccination recommendations by age
Also read:
For any query or additional information, please leave a message below this article and be assured of a response soon or write to info@drvarsha.com.